Researchers Demonstrate Successful Type 1 Diabetes Treatment in Animal Study
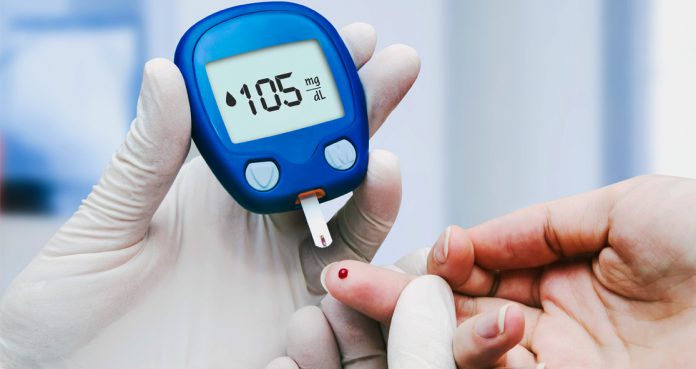
A new study has demonstrated the successful use of type 1 diabetes treatment in an animal model. The study findings suggest that modifying the body’s immune system could help treat type 1 diabetes. The study was published in Science Advances.
Scientists Identify a Potential New Target for Treating Pediatric Blood Cancer
A new study has found a possible new target for treating a type of pediatric blood cancer called juvenile myelomonocytic leukemia. The study identified that dexamethasone, a commonly used anti-inflammatory drug, helped treat juvenile myelomonocytic leukemia in zebrafish.
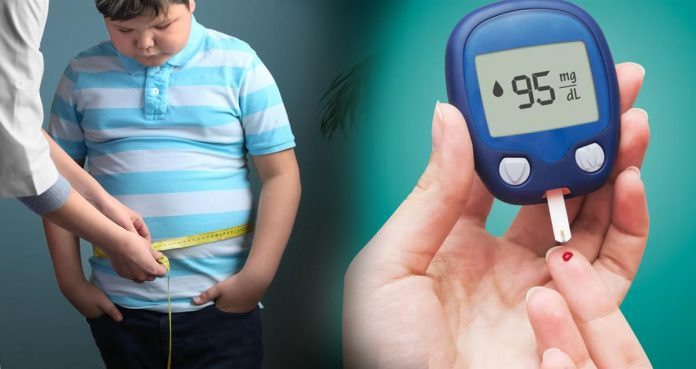
Obese Children Are At Risk of Developing Type 1 Diabetes Later In Life
A new study has shown that obesity in childhood increases the risk of type 1 diabetes in later life, suggesting that childhood obesity is one of the potential reasons why there is an increased prevalence of type 1 diabetes. Also, the study found that childhood obesity also increases the risk of asthma, eczema, and hypothyroidism.
Nitrites and Nitrates Intake Linked To Increased Cancer Risk
A new study has found that people who consume food products containing nitrates and nitrites are more likely to develop cancer. The study found that people who consumed nitrate-based products had a 25% risk of developing breast cancer and those who consumed nitrate-based foods had a 60% risk of developing prostate cancer.
Rilzabrutinib Shows Promise in Treating Blood Dyscrasia
In Phase 1-2 trial, rilzabrutinib has shown promising safety and efficacy in treating a blood disorder called immune thrombocytopenia, which is characterized by low levels of the blood cells that prevent bleeding (platelets). A major Phase 3 trial is currently underway.
Elderly with Cardiovascular Risk Factors Likely to Have Depression, Finds Study
A new study has shown that older individuals with high or very high cardiovascular risk are more likely to have depression. The study was published recently in the journal PLOS One. Cardiovascular disease and depression have often been interlinked because they share similar risk factors.
Entry of Cheaper Generic Versions of Jardiance and Tradjenta Temporarily Blocked In India
In India, the entry of cheaper versions of two commonly used diabetes medicines – Jardiance and Tradjenta – has been temporarily blocked, denying patients affordable drugs. Jardiance and Tradjenta are used to treat type 2 diabetes.