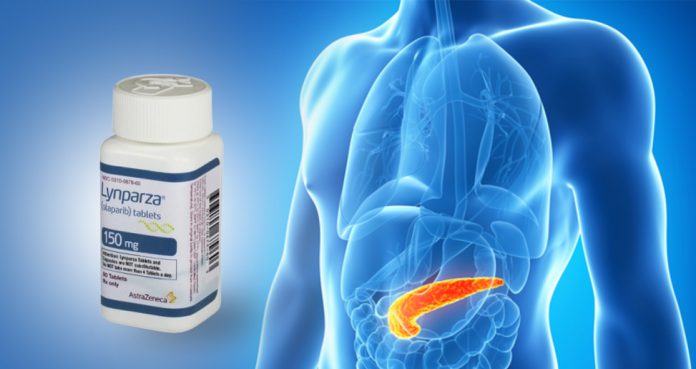
The U.S. Food and Drug Administration (FDA) has approved Lynparza (olaparib) as maintenance therapy for patients with pancreatic cancer.
Developed by AstraZeneca and Merck, Lynparza has been approved as a maintenance treatment of metastatic pancreatic adenocarcinoma in adult patients, whose disease has “not progressed on at least 16 weeks of a 1st-line platinum-based chemotherapy regimen.”
In a joint statement, AstraZeneca and Merck said, “Patients will be selected for therapy based on an FDA-approved companion diagnostic for Lynparza.”
The FDA approved Lynparza after reviewing the results from its clinical trial that showed a significant and clinically meaningful improvement in pancreatic cancer as first-line maintenance therapy.
Pancreatic cancer is a disease with a low survival rate as the disease is diagnosed in its late stage. Worldwide, there were more than 460,000 newly diagnosed cases in 2018. More than 80 percent of those cases are detected when cancer has metastasized, making it difficult to treat.
Lynparza is a PARP inhibitor that has been approved for ovarian and breast cancers.
The head of AstraZeneca’s oncology business unit, Dave Fredrickson, said, “Patients with advanced pancreatic cancer historically have faced poor outcomes due to the aggressive nature of the disease and limited treatment advances over the last few decades.”
“Lynparza is now the only approved targeted medicine in biomarker-selected patients with advanced pancreatic cancer,” he added.
Roy Baynes of Merck Research Laboratories said, “The expanded approval of Lynparza represents a ‘significant milestone’ for patients and also supports the value of germline BRCA testing in patients with this disease.”
Lynparza’s approval emphasizes the increasing importance of PARP inhibitors in treating cancers. In fact, a recent study has found that PARP inhibitors could be effective in treating ovarian and prostate cancer. Another study found that PARP inhibitors, in association with DNA methyltransferase (DNMT) inhibitor, could help in treating some lung cancers.