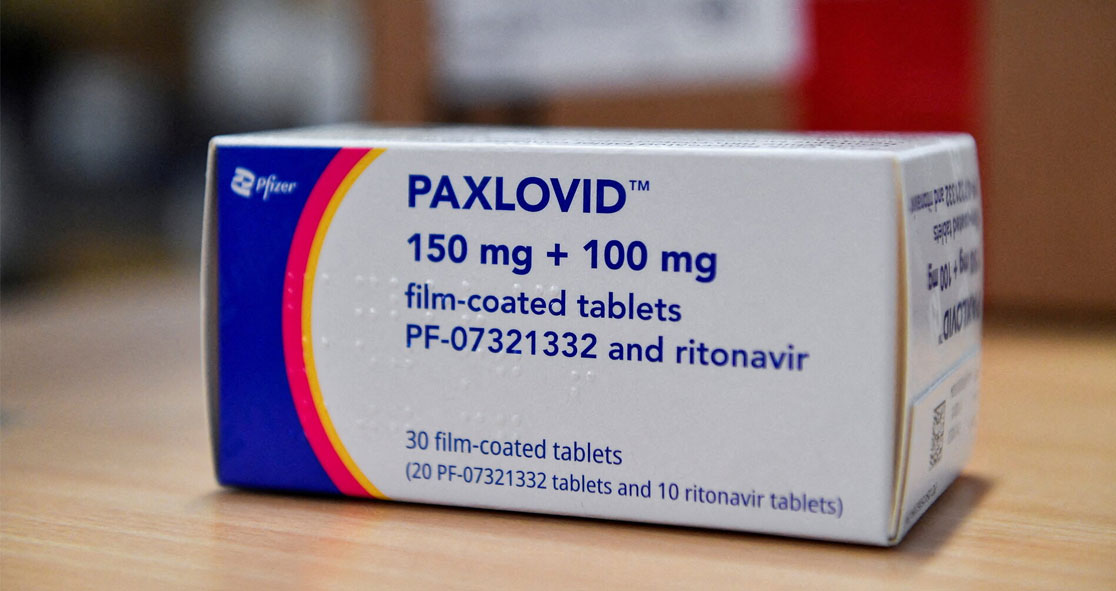
In India, doctors are hopeful that the generic version of Pfizer’s Paxlovid (nirmatrelvir and ritonavir) will be available this week, according to Business Standard.
The drug is likely to play a crucial role if the number of COVID-19 cases rises in India due to the new Omicron descendant.
Hetero Drugs, a Hyderabad-based pharmaceutical company, has already received approval from the Drugs Controller General of India (DCGI) to launch generic Paxlovid in India on April 21, with sources indicating that it may be available this week across the nation.
Last month, the UN-backed Medicines Patent Pool (MPP) signed a license agreement with 35 companies to manufacture the generic version of Paxlovid and supply it to 95 low-and middle-income countries.
Apart from Hetero, the generic version of Paxlovid will be developed by Torrent, Cadila, Biocon, Strides, Glenmark, Granules, Macleods, Sun Pharma, Cipla, among others.
Doctors are optimistic about the drug’s availability as the number of COVID-19 cases in India is rising due to BA.2.12.1 – an Omicron sub-lineage.
Dr. Praveen Gupta, Principal Director of Neurology, Fortis Memorial Research Institute, said, “The introduction of Paxlovid in India is a very welcome addition to the COVID-19 drugs arsenal.”
“The studies have shown that this is a powerful agent in non-hospitalized patients in the early stages of COVID-19 to reduce the risk of hospitalization or death by 89%,” he added. “This is one of the most powerful oral therapies now available to combat COVID-19 infections.”
Dr. Gupta, however, cautioned that Paxlovid has shown to have some drug interactions. He said, “Therefore, one must consult their doctor before taking this medicine, especially the ones who are on multiple concurrent medicines.”
Dr. Rahul Pandit of Fortis Hospital in Mumbai said that generic Paxlovid will be a useful drug for COVID-19 patients.
Pfizer’s Paxlovid is advised as three tablets – two pills of nirmatrelvir and one pill of ritonavir – taken together twice daily for five days. The drug should be started soon after the diagnosis of COVID-19 or within five days of symptom onset.
The World Health Organization (WHO) has “strongly recommended” Paxlovid for patients with mild to moderate COVID-19 who are at risk of getting hospitalized.
The WHO says, “Pfizer’s oral antiviral drug (a combination of nirmatrelvir and ritonavir tablets) is strongly recommended for patients with non-severe COVID-19 who are at highest risk of hospitalization, such as unvaccinated, older, or immunosuppressed patients, with lack of vaccination as an additional risk factor to consider.” Paxlovid has been recommended over Merck’s molnupiravir, another antiviral COVID-19 pill, and Gilead’s remdesivir (Veklury).