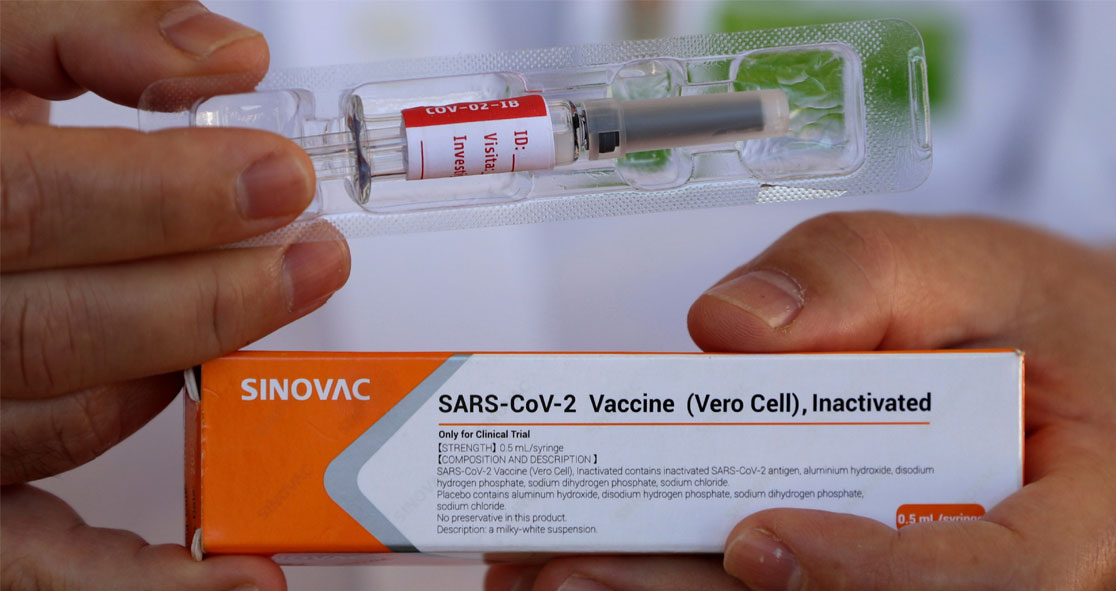
Preliminary results of a COVID-19 vaccine candidate called CoronaVac, developed by China’s Sinovac Biotech, appeared to be safe in a Phase III clinical trial in Brazil.
Brazil’s top biomedical research center called São Paulo’s Butantan Institute said CoronaVac has been proved safe so far in the late-stage trial that involves 9,000 volunteers.
However, Dr. Dimas Covas, head of Butantan, said data on the vaccine’s efficacy will not be released until the trial is completed on all of the 13,000 volunteers.
João Doria, Governor of São Paulo, told reporters, “The first results of the clinical study conducted in Brazil prove that among all the vaccines tested in the country, CoronaVac is the safest, the one with the best and most promising rates.”
Dr. Covas said, “The result is only preliminary and researchers will keep monitoring the participants in the on-going trial.” He also said there were no severe side effects reported with the vaccine so far.
Jean Gorinchteyn, São Paulo State Health Secretary, said CoronaVac appeared to produce protective antibodies.
The state hopes to get regulatory approval for CoronaVac by December to start inoculating its population early in 2021.
São Paulo has signed a deal with Sinovac Biotech to buy 60 million doses of CoronaVac by the end of February.
Sinovac wanted to conduct large trials in Brazil because it has been one of the global hotspots of the coronavirus. Brazil has so far reported more than 5.2 million coronavirus cases and over 154,200 deaths.
Meanwhile, competing coronavirus vaccine candidates, especially the ones developed by AstraZeneca and Johnson & Johnson, have had their trials suspended in the United States over safety concerns.
Experts say vaccines are an essential tool to curb the ongoing pandemic, which has so far killed more than 1 million people globally.
According to Sinovac, CoronaVac uses inactivated vaccine technology, a conventional way of using a killed virus, which cannot replicate in human cells to cause an immune response.
Scientists have warned that the safety of such vaccine candidates must be evaluated over longer periods. They say some other inactivated vaccines had led to infection-enhancement effect, where vaccine-triggered antibodies could worsen the infection when people are exposed to a virus after inoculation, instead of offering protection.