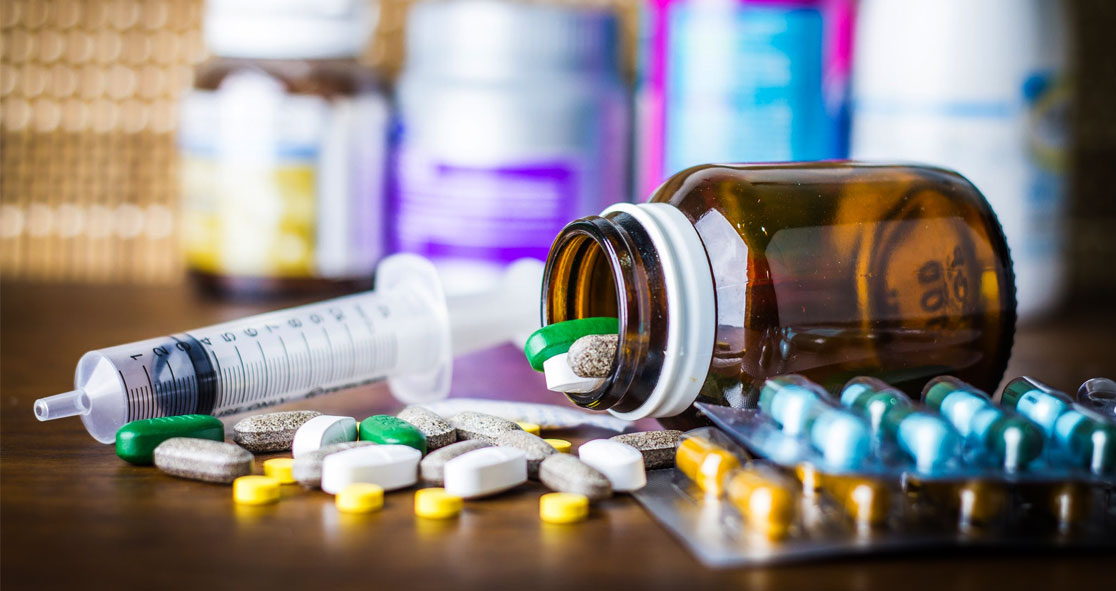
Millions of Americans have been affected by high prescription drug prices, making it difficult for them to pay for their prescriptions. So, they need generic drugs.
Generic drugs are nothing but copies of their brand-name counterparts. They are bioequivalent in terms of the active ingredients, dosages, safety, strength, route of administration, quality, and indications. Simply put, a generic drug works as same as its brand-name version.
And to help Americans have access to cheaper generic medicines, the U.S. Food and Drug Administration (FDA) has placed a high priority on working to ensure a supply of safe, effective, and cost-saving generic drugs, each manufactured under our rigorous quality standards, according to Dr. Janet Woodcock, Director, FDA’s Center for Drug Evaluation and Research.
Generic drugs approved by the FDA have accounted for 90% of the prescriptions dispensed in the nation. In the last decade or so, generic drugs and biosimilars have saved the U.S. health care system billions of dollars.
“The FDA Generic Drugs Program conducts a rigorous review to make sure generic medicines meet these requirements,” according to the agency’s website.
Every year, the agency conducts 3,500 inspections of generic drug manufacturing plants, ensuring compliance with the FDA’s regulations on good manufacturing practices (GMP).
The success of the FDA Generic Drug Program, coupled with recent efforts such as our Drug Competition Action Plan, “has often been described in terms of our ability to efficiently determine the safety and efficacy of generic drugs under review for approval and how many new generics we subsequently approve as a result,” says Dr. Woodcock.
“Safety, effectiveness, and quality are all vital components to the success of our program,” she adds.
At the same time, the FDA continually monitors generic drugs to ensure they are safe and effective at all levels of the supply chain, from active pharmaceutical ingredients (API) to the drugs being sold to consumers.
Like brand-name drugs, generic drugs must meet high standards to receive FDA approval. The agency requires manufacturers to demonstrate that their generic medicines can be effectively substituted with their brand-name counterparts. Also, they should provide the same clinical benefit as the brand-name versions.
Generic drug manufactures must submit the abbreviated new drug application (ANDA) to the FDA, which must show that their medicines are as same as the brand-name version in terms of:
- Active ingredient(s)
- Strength
- Uses or indications
- Dosage form
- Route of administration
If the inactive ingredients of a generic drug are different, it is acceptable. The FDA strictly required a generic medicine to be manufactured under the same strict standards as the brand-name medicine.
For more information about generic drug quality control, check with your doctor, pharmacist, or other health care provider. For more information, you can also: You can also visit the FDA Generic Drugs Program web page to know more about the quality control of generic drugs in the United States.